Corrosion control of carbon steel heat exchange components is becoming increasingly more important as energy costs rise and the demand for environmentally friendly chemicals is on the water technology forefront. This series of articles explores the effectiveness of both inorganic and organic inhibitors to control carbon steel corrosion in open system cooling tower water that is both corrosive and scaling. Pilot-scale corrosion testing is used to generate results from corrosion coupons and corrator electrodes under synthetic water conditions. Several carbon steel corrosion inhibitors are examined. Carbon steel corrosion inhibitor packages are discussed and compared in terms of environmental friendliness, corrosion rate minimization, and cost effectiveness.
INTRODUCTION
Metals have been used for many years to minimize carbon steel corrosion. Inorganic compounds such as chromate, zinc, and molybdate, often times combined with phosphate or polyphosphate, are added to aqueous systems to provide corrosion protection for carbon steel. Prevention of costly metal loss due to general corrosion and pitting is accomplished by the use of these anodic and cathodic inhibitors. In the last several years, these materials have been regulated more stringently by the Environmental Protection Agency (EPA) and other governing agencies due to their perceived negative impact on the environment or other safety concerns. For example, hexavalent chromium compounds are considered to be known human carcinogens (see reference 1) and the overall non-biodegradability of the metals is considered undesirable as they tend to accumulate in the environment (see reference 2). Molybdate has come under scrutiny because of the apparent negative response of lactating dairy cows to high levels of dietary molybdenum (see reference 3). Phosphate can form algae blooms that limit the oxygen in lakes and streams from agricultural run-off and other means causing the death of aquatic life (see reference 4). Even though these materials have worked well to prevent expensive corrosion of carbon steel, they are becoming less and less available or the amount that can be used for this purpose has been limited substantially.
In light of these environmental and safety restrictions, newer non-metallic corrosion inhibitors have become commercially available. These organic inhibitors are often based on low molecular weight polymers, phosphonates, and amino phosphonates. Some of these organic molecules have proven to be successful carbon steel corrosion inhibitors (see reference 5). Although they show improved environmental acceptability, they too may have some technical concerns. For instance, organic molecules may be more susceptible to oxidizing biocides than inorganic molecules. Many azole-based copper corrosion inhibitors are susceptible to degradation by oxidizing biocides. Hydroxyphosphonic acid has shown susceptibility to chlorine, even at low levels (see reference 5). This presents a problem because oxidizing biocides are a necessary part of any successful water technology program for control of potentially harmful bacteria, such as legionella. Organic inhibitors based on the phosphate molecule may result in unexpected phosphorous in the open cooling system due to reversion of some of the organic phosphonate to orthophosphate. Reversion of the organic phosphorous molecule may render it less effective as an inhibitor but may be a good biological food source. Finally, organic inhibitors can be deactivated by cationic microbiocides such as the quaternary ammonium compounds and other cationic substances that may be present (see reference 6).
It should be noted that tin is one inorganic inhibitor recently available that is less regulated than other metals. It can be used in drinking water, carries some Food and Drug Administration (FDA) accepted food uses, is considered quite safe for the environment (see reference 7), and for human safety in general (see reference 8). Tin used for corrosion inhibition in cooling towers currently has patents associated with its use (see reference 9).
Green Chemistry has become a design criterion in water treatment programs and the chemical industry over the past decade. The definition of Green Chemistry is constantly evolving to include new environmental regulations and customer expectations. In general, it may be assumed that Green Chemistry is environmentally friendly, but may have other connotations that apply to safety, good manufacturing practice, and even efficiency.
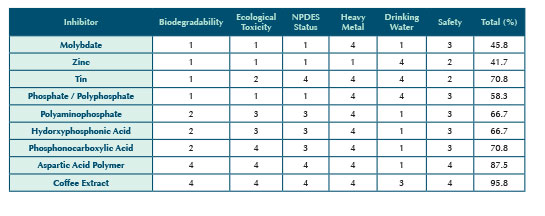
GREEN CHEMISTRY CRITERIA
In our pursuit of a green carbon steel corrosion inhibitor, we strive to be as green as possible, and yet perform adequately for the purpose intended. For the purpose of these articles, the inputs that are used to determine green status for an inhibitor fall into six categories. A summary of the evaluation is shown in Table 1. The values are the subjective interpretation by the authors.
Biodegradability
Inorganic materials are not usually considered biodegradable but instead they tend towards bioaccumulation, implying a negative situation, thus their green rating for this criterion is low. An organic chemical substance is said to be biodegradable if it can be broken down by the metabolic action of living microorganisms such as bacteria, fungi, algae, and yeast. Biodegradation is one of the most important processes governing the fate of a chemical after its release into the environment. A highly biodegradable material is considered greener than more persistent counterparts.
Ecological Toxicity
Toxicity testing can be done to determine the effects of a material on aquatic species like fish, water flea, and algae. The less ecologically toxic a material is, the greener its status is considered to be.
NPDES Status
The National Pollution Discharge Elimination System (NPDES) issues permits regulating chemical discharge for the protection of the environment. The materials that are regulated on these permits and the concentrations allowed for discharge to the environment will affect the green ranking of a material.
Heavy Metal Status
The EPA has established a list of heavy metals, also called Target Analyte Metals, that are used to determine environmental assessments such as NPDES permitting. Materials listed as Target Analyte Metals are considered less green than others.
Drinking Water Additive
Certain additives are allowed by the EPA as corrosion inhibitors in drinking water systems. These normally carry a maximum allowed use level. These materials usually carry a third party certification from the National Sanitation Foundation (NSF) confirming they are safe for use in drinking water. A drinking water additive could be considered greener than materials that cannot be added to drinking water.
Safety
Materials are rated for safety by several factors: the Occupation Safety and Health Administration (OSHA) through a Material Safety Data Sheet, the United States Department of Transportation (USDOT) through the Hazardous Materials Table, the FDA through a Generally Regarded as Safe (GRAS) status, and others sources as they apply. A higher safety rating translates to a better green status.
CHEMICAL INHIBITORS
The additives chosen for our pilot testing are common inhibitors that are available in industry. The list is not exhaustive but gives a fair representation of the kinds of technology currently available. Descriptions of various corrosion inhibitors can be found in the AWT Technical Reference and Training Manual (see reference 5).
Molybdate
Molybdate is a precipitation inhibitor at anodic sites of carbon steel. Molybdate is often combined with other corrosion inhibitors. The molybdate source was sodium molybdate dihydrate.
Zinc
Zinc inhibits corrosion of carbon steel by forming hydroxide or hydroxycarbonate compounds on the surface. Zinc is often combined with other corrosion inhibitors. A solution of zinc chloride was used.
Tin
The mechanism for tin corrosion inhibition of carbon steel is not well understood. A 50 percent tin chloride solution was used. The use of this material as a corrosion inhibitor in cooling towers is patented (see reference 9).
Orthophosphate
Orthophosphate forms ferric phosphate at anodic sites of carbon steel forming a protective film. Both monosodium phosphate (MSP) and disodium phosphate (DSP) salts were used.
Polyphosphate
The mechanism of polyphosphate inhibition is complex, but can be generalized by precipitation at cathodic carbon steel sites. Polyphophate is often combined with other corrosion inhibitors. The polyphosphate source was tetrapotassium pyrophosphate (TKPP).
Polyaminophosphonate
Polyaminophosphonate (PAP) is a phosphonate inhibitor that precipitates a general protective film at both anodic and cathodic sites on carbon steel. A liquid solution of PAP was used.
Hydroxyphosphonic Acid
Hydroxyphosphonic acid (HPA) is a phosphonate inhibitor that precipitates a general protective film at both anodic and cathodic sites on carbon steel. A liquid solution of HPA was used.
Phosphonocarboxylic Acid
Phosphonocarboxylic acid (PCA) works similarly to HPA as a corrosion inhibitor, but is claimed to have higher resistance to chlorine degradation. A liquid solution of PCA was used.
Aspartic Acid Polymer
The mechanism for aspartic acid polymer (AAP) inhibition of carbon steel corrosion is not well known. Manufacturer testing has claimed carbon steel corrosion inhibition under certain synthetic brine conditions. A liquid solution of AAP was used.
Coffee Extract
It is unknown which chemical components of coffee extract directly contribute to carbon steel corrosion inhibition (see reference 10). A viscous coffee extract material was used in the experiments.
A LOOK AHEAD
In next month’s installment in this series, we will detail the experimental procedure’s test apparatus, with controlled flow rates and temperature to simulate both a scaling and corrosive environment. Later installments will build on these observations to determine acceptable green corrosion inhibitors for use in open system cooling waters by examining the effect of oxidizing biocide on their performance and, finally, to select the most appropriate and cost effective product for use as a green corrosion inhibitor. ■
REFERENCES
- Report on Carcinogens, Twelfth Edition, National Toxicology Program, Department of Health and Human Services, 2011. Web. May 17, 2012.
- Qishlaqi and F. Moore, “Statistical analysis of Accumulation and Sources of Heavy Metals Occurrence in Agricultural Soils of Khoshk River Banks, Shiraz, Iran”, American-Eurasian J. Agriculture and Environmental Science, 2, 565-573 (2007).
- J.T. Huber, N.O. Price, and R.W. Engel, “Response of Lactating Dairy Cows to High Levels of Dietary Molybdenum”, Journal of Animal Science, 32, (1971).
- B. Oram, Water Research Center, B. F. Environmental Consultants Inc., 2012. Web. May 17, 2012. “Phosphates in the Environment.”
- Association of Water Technologies Technical Reference and Training Manual, 2nd ed, chapter 5.10, (2009).
- P. Puckorius et al., “Phosphonates-Part II-Corrosion Control- Cooling Water”, A Practical Guide to Water Treatment Chemicals Newsletter, Volume 2, Issue 1, 1st quarter (1998).
- P. Howe, M. Wood, and P. Watts, “Tin and Inorganic Tin Compounds”, Concise International Chemical Assessment Document, 65, World Health Organization Geneva, (2005).
- J. Fawell and R. Mascarenhas, “Inorganic Tin in Drinking-water”, Background document for development of WHO Guidelines for Drinking-water Quality, World Health Organization, (2004).
- W.E. Stapp and G. Westlund, “Corrosion Inhibition Compositions and Methods for using the same”, U.S. Patent 7,910,024, issued March 22, 2011.
- V.V. Torres et al., “Inhibitory action of aqueous coffee ground extracts on the corrosion of carbon steel in HCl solution”, Corrosion Science, 53, 2385-2392 (2011).
- B.P. Boffardi, “Corrosion and Fouling Monitoring of Water Systems”, The Analyst Technology Supplement, spring (2010).
- Rohrback Cosasco Systems, Inc., “Model 9020 &9020-OEM Corrater Transmitter User Manual”, November (2004).
About the Author
Matthew LaBrosse, PhD, is an engineer in research and development at U.S. Water Services, Inc. in St. Michael, Minnesota, and can be reached at mlabrosse@uswaterservices.com. Donovan Erickson is a certified water technologist for U.S. Water Services, Inc. The authors wish to thank the Association of Water Technologies for allowing this series to be presented and to U.S. Water Services for the resources necessary to conduct the research. For more information, visit www.uswaterservices.com.
MODERN PUMPING TODAY, January 2015
Did you enjoy this article?
Subscribe to the FREE Digital Edition of Modern Pumping Today Magazine!


